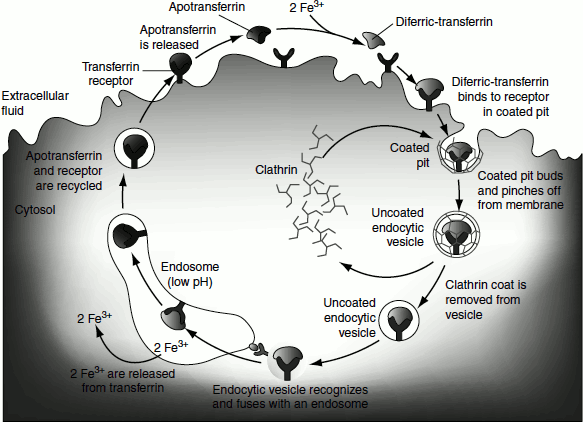
Some proteins such as low-density lipoprotein (LDL), the iron-carrying protein transferrin, peptide hormones and circulating proteins, which are destined to be degraded, are imported into certain cells from the surrounding medium. These proteins bind to receptors on the outer face of the plasma membrane. These receptors are concentrated in the invaginations of the membrane called coated pits, which are coated on their cytosolic side with a lattice made up of the protein called clathrin.
Clathrin forms closed polyhedral structures. Clathrin is a trimer of three light (L) chains and three heavy (H) chains. The (HL)3 clathrin unit is organized as a three-legged structure called a ‘triskelion’. Triskelions have a propensity to assemble into polyhedral lattices (Figure 6.23).
As more of the receptors become occupied with target proteins, the clathrin lattice grows until a complete membrane-bounded endocytic vesicle buds off the plasma membrane and moves into the cytoplasm.
The clathrin is quickly removed by uncoating enzymes and the vesicles fuse with endosomes. The pH of endosomes is lowered by the activity of V-type ATPases in their membranes. This creates an environment that facilitates the dissociation of receptors from their target proteins. Proteins and receptors then go their separate ways.
Receptor-mediated endocytosis (Figure 6.24) is exploited by some viruses to gain entry to cells. Influenza virus enters cells this way. HIV, the virus that causes AIDS, also binds to specific receptors on the cell surface and may gain entry by endocytosis. In humans, the receptor that binds HIV, known as CD4, is a glycoprotein found primarily on the surface of immune system cells called helper T-cells. CD4 is normally involved in the complex communication between cells of the immune system that is required to execute the immune response.
Figure 6.23 Clathrin
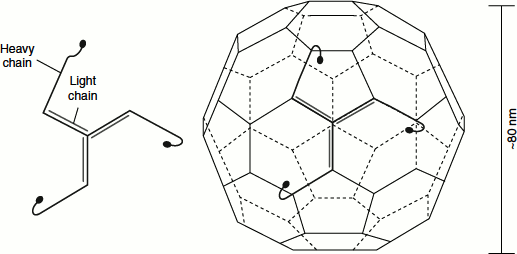
Figure 6.24 Clathrin-mediated enocytosis