Chemical Mutagens
Mutations can be caused by chemicals such as:
- Base analogues,
- Chemicals that modify bases,
- Intercalating agents and
- Alkylating agents.
Base analogues
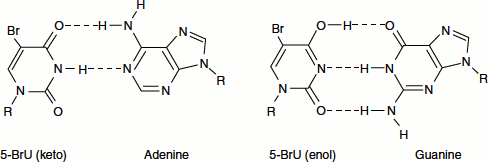
Compounds that resemble the four bases of nucleic acids are called base analogues for example, 5-bromouracil and 2-amino purine. These base analogues can be incorporated into DNA. They lead to transition mutations (purine to purine) as a consequence of altered base pairing in a subsequent round of DNA replication, 5-bromouracil, analogue of thymine, normally pairs with adenine. However, the enol tautomer of the compound is more predominant and pairs with guanine. This causes AT ↔ GC transitions (Figure 8.3).
Figure 8.3 AT → GC transition by 5-bromouracil
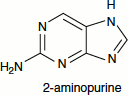
2-Aminopurine is an analogue of guanine and adenine. It commonly pairs with thymine as an adenine analogue and can also pair with cytosine as a guanine analogue. Thus, it can produce an AT ↔ GC transition.
Chemicals that modify the bases of the DNA
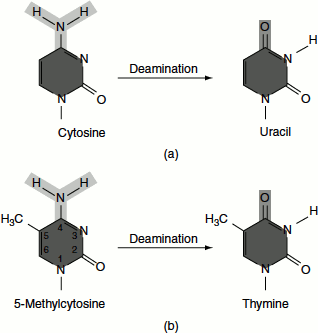
Certain mutagens act by chemically modifying the bases; for example, nitrous acid converts amino groups to keto groups by oxidative deamination. Adenine is oxidatively deaminated to hypoxanthine, cytosine to uracil and guanine to xanthine.
Cytosine → deamination → uracil → pairs with Adenine; CG → UA transition
Adenine → deamination → hypoxanthine → pairs with C; AT → GC transition.
Guanine → deamination → xanthine → pairs with C.
Hydroxyl amine (NH2OH) is another mutagen that specifically reacts with cytosine and converts it into a modified base that pairs only with A.
Intercalating agents
Flat aromatic molecules such as acridines act as mutagens by intercalating in the DNA, i.e., they slip in-between adjacent base pairs in the DNA and consequently lead to the insertion or the deletion of one or more base pairs. This alters the reading frame; for example, proflavine and acridine orange (Figure 8.4).
Alkylating agents
These add alkyl groups to the hydrogen bonding oxygen of guanine and thymine producing O-6-alkyl guanine, which pairs with thymine, and O-4-alkyl thymine, which pairs with guanine.
Figure 8.4 Intercalating agents

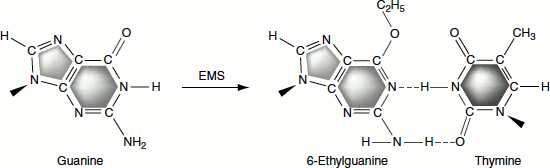
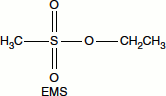
Ethylmethane sulphonate (EMS), also known as mustard gas, is an alkylating agent that can donate (C2H5) group to a keto group in guanine, resulting in the formation of 6-ethylguanine that acts as an analogue of adenine and pairs with thymine, resulting in GC → AT transitions.
Physical Mutagens
‘Ultraviolet light’ is a potent mutagen. The main DNA damages caused by UV light include cyclobutane-pyrimidine dimmers and pyrimidine dimmers (Figure 8.5).
‘Ionizing radiations’ such as X-rays and gamma rays emitted by radioactive material cause breaks in the phosphodiester bonds of the DNA and result in chromosome mutations such as inversions, translocation and duplications. Radiations can also cause ionizations or ring opening in bases or they can open sugar rings or break strands and even they can cause chromosomal breaks. The radiations that cause the ionization of bases can lead to wrong base pairing. Natural sources of radiation produce so-called background radiation. These include cosmic rays from the Sun and outer space, radioactive elements in soil and terrestrial products (wood, stone, etc.) and in the atmosphere (radon) (Figure 8.6).
Biological Agents of Mutation
There are several biological agents that can cause DNA mutation. This may include viruses and bacteria. Some of the viruses donate their DNA materials to the host cell and cause alteration in the genetic code (transposons). The viruses that may cause DNA mutations include human papilloma virus (implicated in cervical cancer), the human T-cell lymphyocytic virus (implicated in lymphoma) and the hepatitis B virus (implicated in liver cancer). Helicobacter pylori is a bacterial agent that may bring about DNA mutation.
Test for Mutagenicity
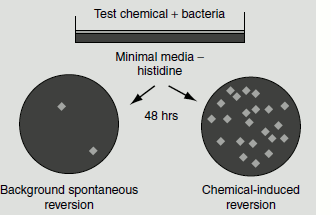
A large number of mutagens can be tested for mutagenicity using nutritional mutants of bacteria. In a simple test, a known number of bacterial mutants are plated on a growth medium containing a potential mutagen and the number of bacterial colonies that reverted back is counted. If the substance is a mutagen, the number of colonies will be greater than that would be obtained in the absence of the mutagen.
Figure 8.5 Pyrimidine dimerization induced by UV light

Figure 8.6 Physical mutagens

These tests, however, fail to demonstrate the mutagenicity of many carcinogens, as these substances are not directly mutagenic. They are converted to actively mutagenic compounds by enzymatic reactions in the liver, which is not seen in bacteria. Hence, the addition of the microsomal fraction to the bacterial growth medium allows the mutagens to undergo enzymatic activation and would enable to determine the mutagenicity of these substances. This is the basic principle employed in ‘Ames test’, which is used to screen potential mutagens/carcinogens.
Figure 8.7 Ames test
Ames test
In this test, histine-requiring (His–) mutants of the bacterium Salmonella typhimurium, which contain either a base substitution or a frame shift mutation are used to test for reversion to His+. The frequency of spontaneous revertants is low in this mutant; however, they are readily produced in the presence of a mutagen. The bacterial medium is prepared containing a very small amount of histidine, which is enough to support the growth of individual cells but not for colony formation. A small amount of rat liver extract and about 108 His– mutants are spread on each plate (group A). Distilled water is applied to the plates in the control group that is devoid of the mutagen (group B). About 5-10 colonies usually appear on plates in group B. In presence of a mutagen, many colonies appear in group A and thus the mutagenicity of the added mutagen can be tested. The Ames test has significantly reduced the testing of mutagens on animals (Figure 8.7).