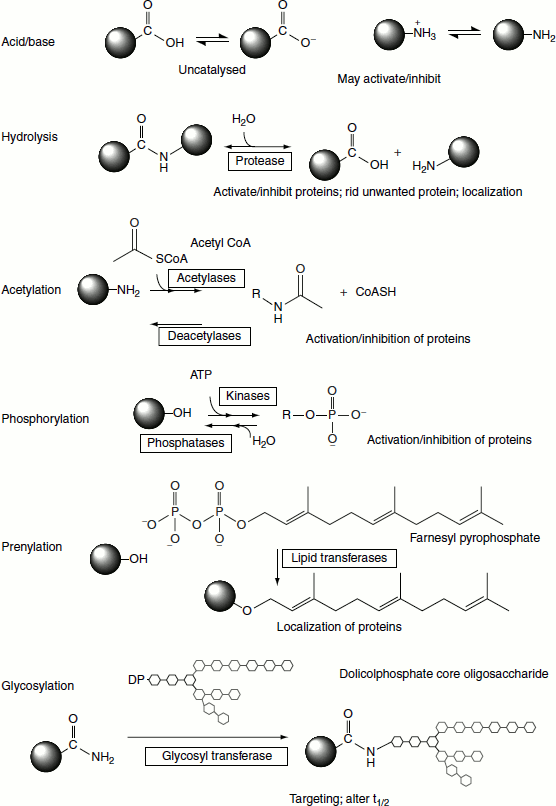
Most of the proteins that are translated from mRNA undergo chemical modifications before becoming functional in different body cells. The modifications collectively are known as post-translational modifications (Figure 5.16). The protein post-translational modifications (PTMs) play a crucial role in generating the heterogeneity in proteins and also help in utilizing identical proteins for different cellular functions in different cell types.
PTM increases the functional diversity of the proteome by the covalent addition of functional groups or proteins, proteolytic cleavage of regulatory subunits or degradation of entire proteins. These modifications include phosphorylation, glycosylation, ubiquitination, nitrosylation, methylation, acetylation, lipidation and proteolysis and influence almost all aspects of normal cell biology and pathogenesis. Therefore, identifying and understanding PTMs is critical in the study of cell biology and disease treatment and prevention.
Proteolytic cleavage
Following translation, most proteins undergo proteolytic cleavage. The simplest form of this is the removal of the initiation methionine. Many proteins are synthesized as inactive precursors that are activated under proper physiological conditions by limited proteolysis. Pancreatic enzymes and enzymes involved in clotting are examples of the enzymes that are activated by proteolytic cleavage. Inactive precursor proteins that are activated by the removal of polypeptides are termed as proproteins.
An example of a preproprotein is insulin. Since insulin is secreted from the pancreas, it has a prepeptide. Following the cleavage of the 24-amino acid signal peptide, the protein folds into proin-sulin. Proinsulin is further cleaved yielding active insulin which is composed of two peptide chains linked together through disulphide bonds.
Still other proteins (of the enzyme class) are synthesized as inactive precursors called zymogens. Zymogens are activated by proteolytic cleavage such as the proteins of the blood clotting cascade.
Acylation
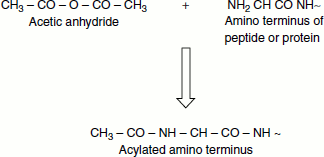
Many proteins are modified at their N-termini following synthesis. In most cases, the initiator methionine is hydrolysed and an acetyl group is added to the new N-terminal amino acid. Acetyl-CoA is the acetyl donor for these reactions.
Myristoylation
N-myristoylation is also an acylation process found to be specific to N-terminal amino acid. Some proteins have the 14-carbon myristoyl group added to their N-termini. The donor for this modification is myristoyl-CoA. The latter modification allows the association of the modified protein with membranes.
Figure 5.16 Post-translational modifications
The catalytic subunit of cyclicAMP-dependent protein kinase (PKA) is myristoylated.The cytosolic enzyme A-myristoyltransferase (NMT) catalyses myristoylation.Myristoylation plays a vital role in the secondary cellular signalling, in the infectivity of retroviruses and oncogenesis in eukaryotes.
Methylation
Post-translational methylation of proteins occurs on nitrogen and oxygen. The activated methyl donor is S-adenosylmethionine (SAM). The most common methylations are on the ε-amine of lysine residues. Additional nitrogen methylations are found on the imidazole ring of histidine, the guanidino moiety of arginine and the R-group amides of glutamate and aspartate. Methylation of the oxygen of the R-group carboxylates glutamate and aspartate and forms methyl esters. Proteins can also be methylated on the thiol R-group of cysteine.
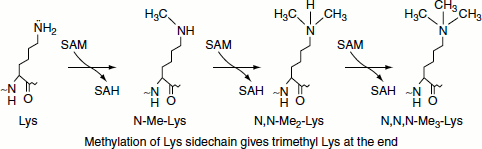
Methylation in the proteins increases the hydrophobicity of the protein. Methylation on carboxylate side chains cover up negative charge and adds hydrophobicity.
For example, N-methylation of lysines does not alter the cationic charge but does increase hydrophobicity.
Phosphorylation
Post-translational phosphorylation is one of the most common protein modifications that occur in animal cells. Phosphorylations regulate the biological activity of a protein and as such are transient. In other words, a phosphate (or more than one in many cases) is added and later removed.
For example, proteins such as glycogen synthase and glycogen phosphorylase in hepatocytes are phosphorylated in response to glucagon release from the pancreas. Phosphorylation of synthase inhib its its activity, whereas the activity of phosphorylase is increased. These two events lead to increased hepatic glucose delivery to the blood.
The enzymes that phosphorylate proteins are termed as kinases and those that remove phosphates are termed as phosphatases. Protein kinases catalyse reactions of the following type:
ATP + protein ↔ phosphoprotein + ADP
In animal cells, serine, threonine and tyrosine are the amino acids that subject to phosphorylation. The largest group of kinases are those that phosphorylate either serines or threonines and as such are termed as serine/threonine kinases. The ratio of phosphorylation of the three different amino acids is approximately 1,000/100/1 for serine/threonine/tyrosine.
Although the level of tyrosine phosphorylation is minor, the importance of phosphorylation of this amino acid is profound.
Acetylation
The acetylation and deacetylation take place on lysine residues in the A-terminal tail in histone acetylation and deacetylation. These reactions take place in the presence of the enzymes histone acetyltransferase (HAT) or histone deacetylase (HDAC).
Formylation
Formylation is one of the post-translational modifications of the protein, in which a protein is modified by the attachment of formyl group. The most commonly studied mechanism is the N6-formylation of lysine. Histone and other nuclear proteins are modified by formylation. The post-translational modification of histone and other chromatin proteins regulates gene expression.
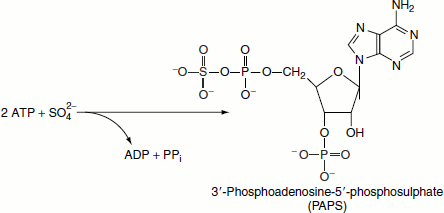
Sulphation
Sulphate modification of proteins occurs at tyrosine residues such as in fibrinogen and in some secreted proteins (e.g., gastrin). The universal sulphate donor is 3′-phosphoadenosyl-5′-phosphosulphate (PAPS).
Prenylation
Prenylation refers to the addition of the 15-carbon farnesyl group or the 20-carbon geranyl group to acceptor proteins, both of which are isoprenoid compounds derived from the cholesterol bioszynthetic pathway. The isoprenoid groups are attached to cysteine residues at the carboxy terminus of proteins in a thioether linkage (C-S-C). A common consensus sequence at the C-terminus of prenylated proteins has been identified and is composed of CAAX, where C is cysteine, A is any aliphatic amino acid (except alanine) and X is the C-terminal amino acid. In order for the prenylation reaction to occur, the three C-terminal amino acids (AAX) are first removed. Following attachment of the prenyl group, the carboxylate of the cysteine is methylated in a reaction utilizing SAM as the methyl donor.
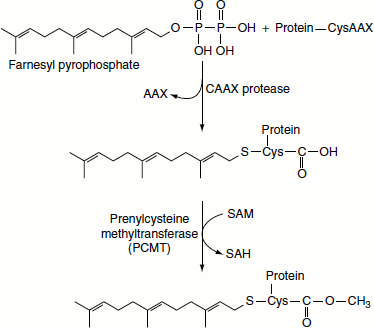
Many proteins are modified at their C-terminus by prenylation near a cysteine residue in the consensus CAAX. Following the prenylation reaction, the protein is cleaved at the peptide bond of the cysteine and the carboxylate residue is methylated by a prenylated protein methyltransferase. One such protein that undergoes this type of modification is the proto-oncogene RAS.
Some of the most important proteins whose functions depend upon prenylation are those that modulate immune responses. These include proteins involved in leukocyte motility, activation and proliferation and endothelial cell immune functions. These immune modulatory roles of many prenylated proteins are the basis for a portion of the anti-inflammatory actions of the statin class of cholesterol synthesis-inhibiting drugs due to a reduction in the synthesis of farnesyl pyrophosphate and geranyl pyrophosphate and thus reduced the extent of inflammatory events. Other important examples of prenylated proteins include the oncogenic GTP-binding and -hydrolysing protein RAS and the γ-subunit of the visual protein transducin, both of which are farnesylated. In addition, numerous GTP-binding and -hydrolysing proteins (termed as G-proteins) of signal transduction cascades have γ-subunits modified by geranylgeranylation.